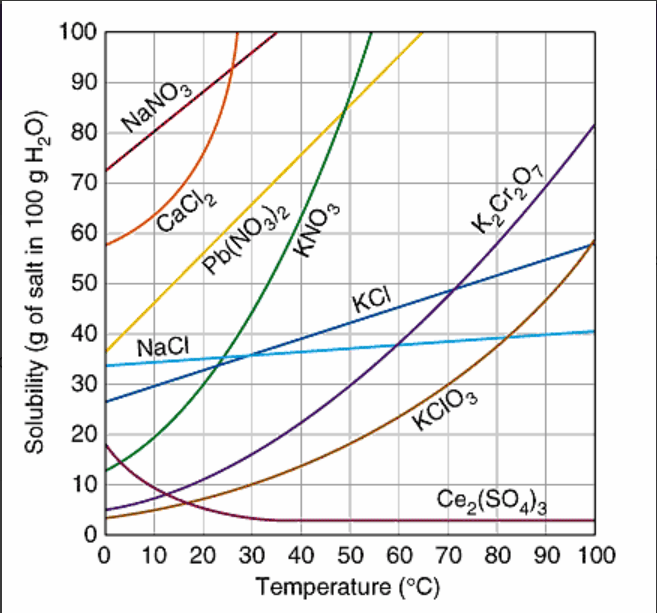
Solubility factors and curve
Assessment
•
Angela Pirtle
•
Science
•
9th Grade
•
27 plays
•
Medium
Improve your activity
Higher order questions
Match
•
Reorder
•
Categorization
.svg)
actions
Add similar questions
Add answer explanations
Translate quiz
Tag questions with standards
More options
15 questions
Show answers
1.
Multiple Choice

At approximately what temperature does the solubility of sodium chloride, NaCl, match the solubility of potassium dichromate, K2Cr2O7?
60oC
30oC
50oC
83oC
2.
Multiple Choice
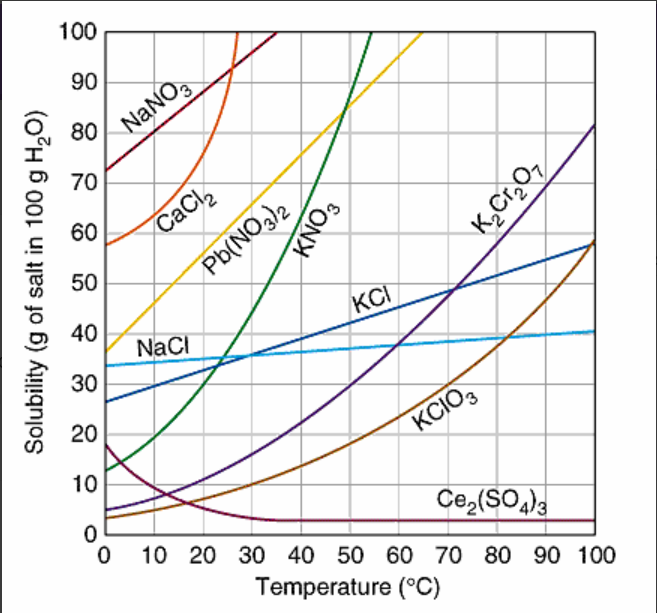
How many grams of K2Cr2O7, are soluble in 100 g of water at 95oC?
83 grams
75 grams
40 grams
12 grams
3.
Multiple Choice
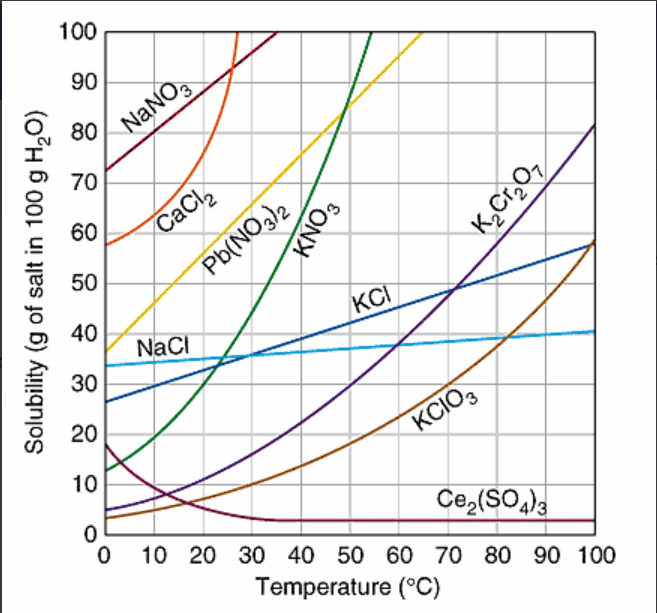
Which substance is MOST soluble at 0oC?
KI
NaNO3
NaCl
Ce2(SO4)3
4.
Multiple Choice
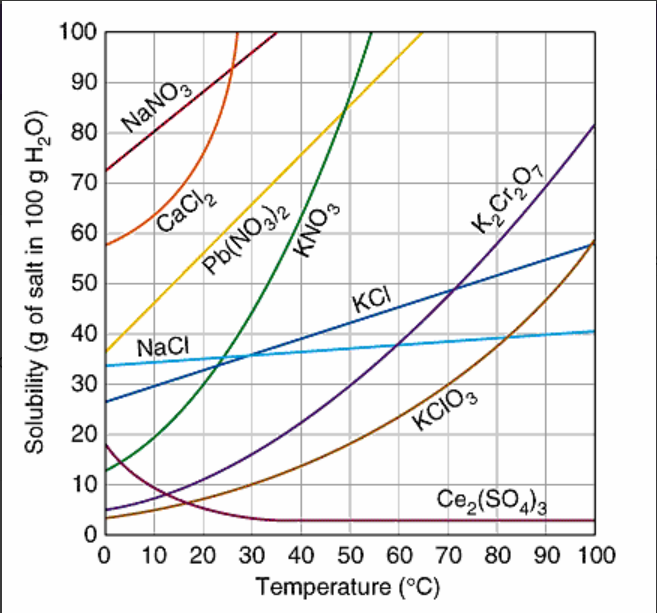
When 20 grams of potassium chlorate, KClO3, is dissolved in 100 g of water at 80oC, the solution can be correctly describe as:
supersaturated
saturated
unsaturated
5.
Multiple Choice
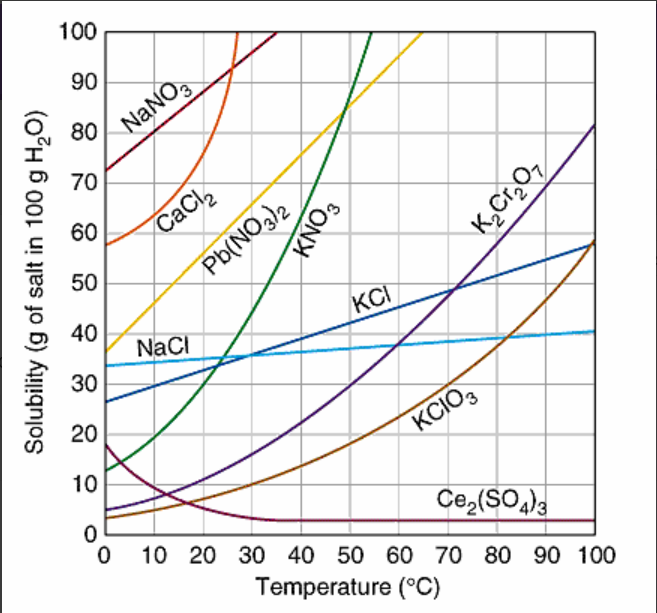
When 42 grams of potassium chloride, KCl, is dissolved in 100 grams of water at 50oC, the solution can be correctly described as:
supersaturated
saturated
unsaturated
6.
Multiple Choice
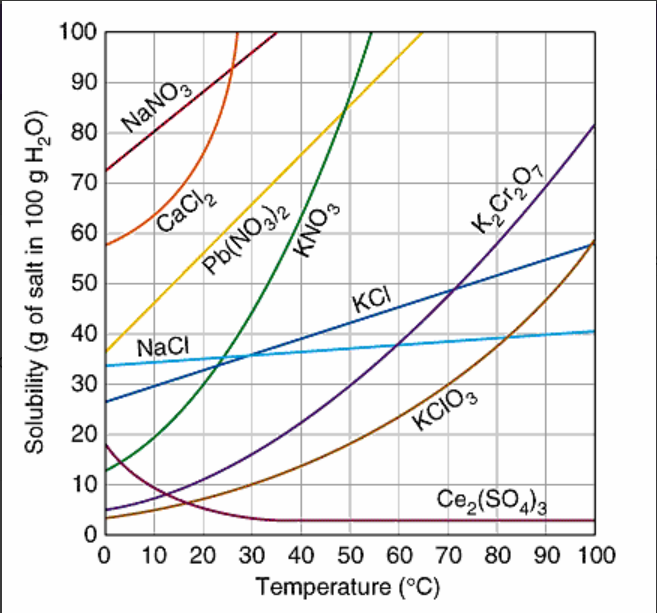
What temperature does the solubility of NaCl match KNO3?
50oC
40oC
35oC
25oC

Explore this activity with a free account
Find a similar activity
Create activity tailored to your needs using
.svg)

Mixtures and Solutions
•
5th Grade

Mixtures and Separating Mixtures
•
4th - 5th Grade

Separating Mixtures
•
7th Grade

Mixtures and Solutions
•
4th - 5th Grade

Properties of Matter
•
3rd - 5th Grade

Mixtures and Solutions
•
5th Grade

Physical Properties of Matter Final
•
5th Grade

Mixtures and Solutions
•
5th Grade