Phases of Water
Assessment
•
Stephanie Molloy
•
Science
•
5th - 8th Grade
•
3 plays
•
Medium
Improve your activity
Higher order questions
Match
•
Reorder
•
Categorization
.svg)
actions
Add similar questions
Add answer explanations
Translate quiz
Tag questions with standards
More options
20 questions
Show answers
1.
Multiple Choice
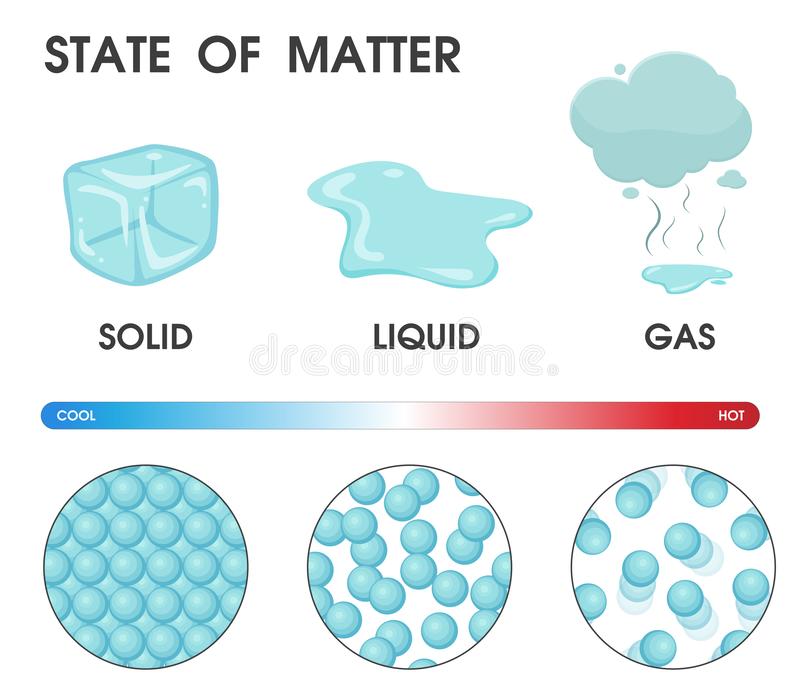
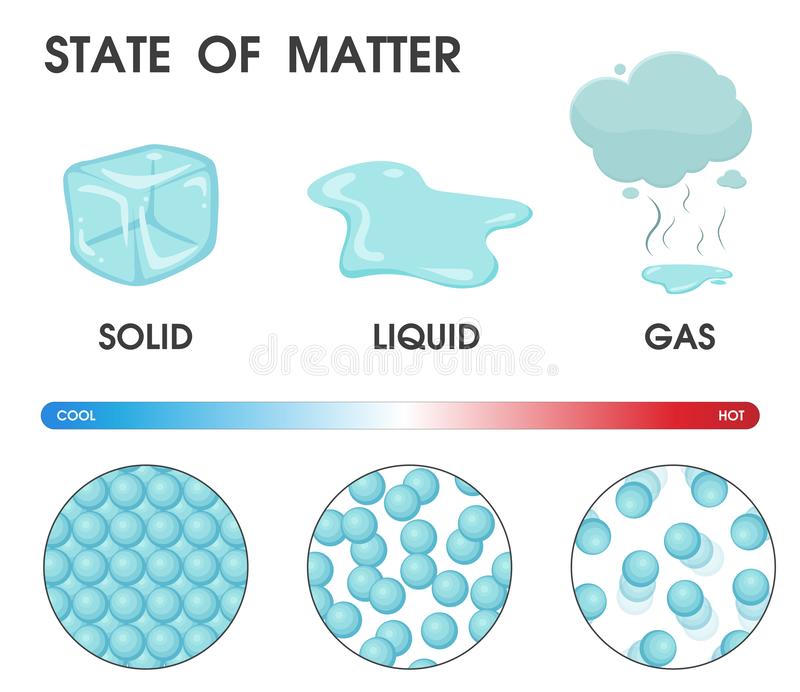
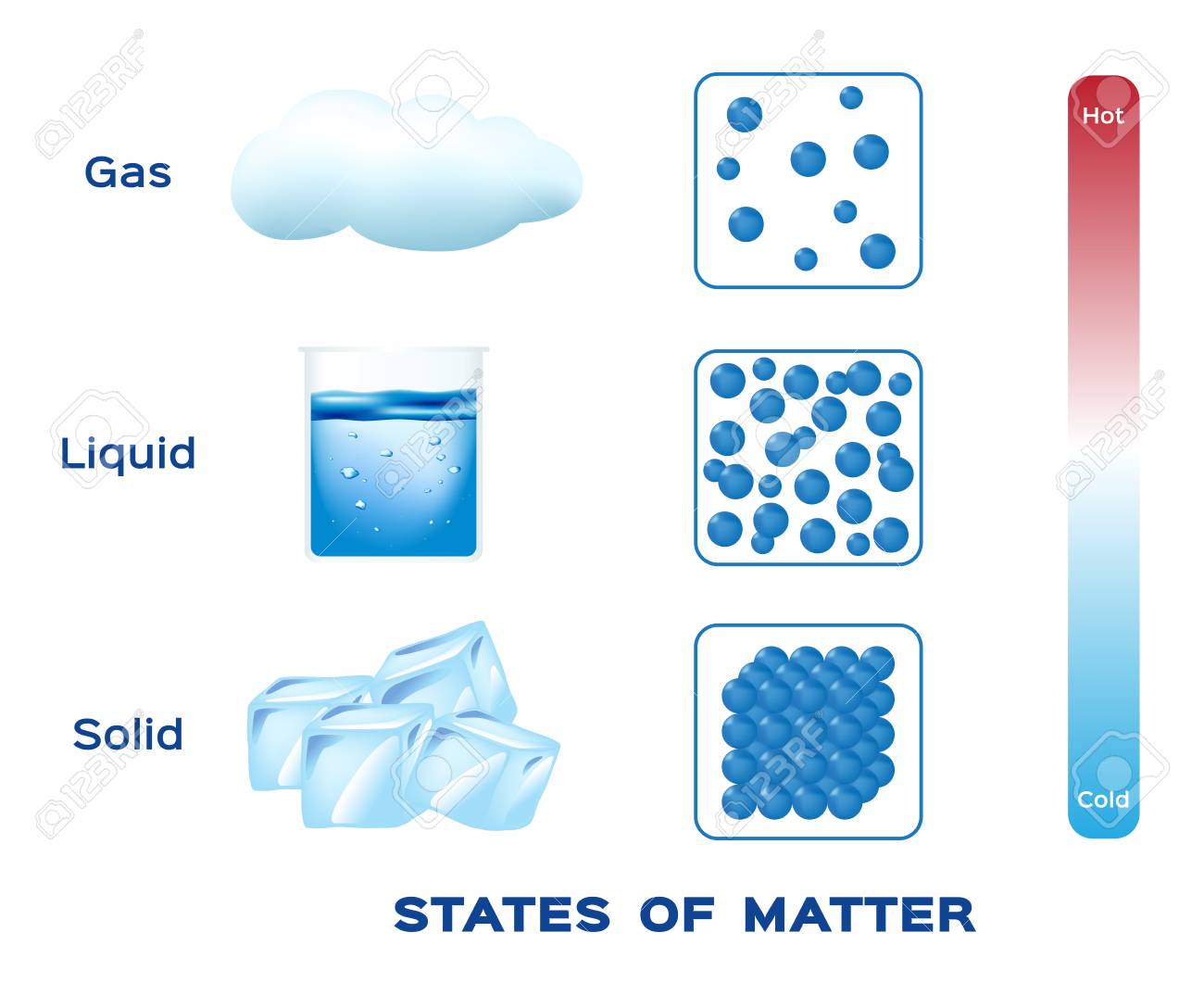
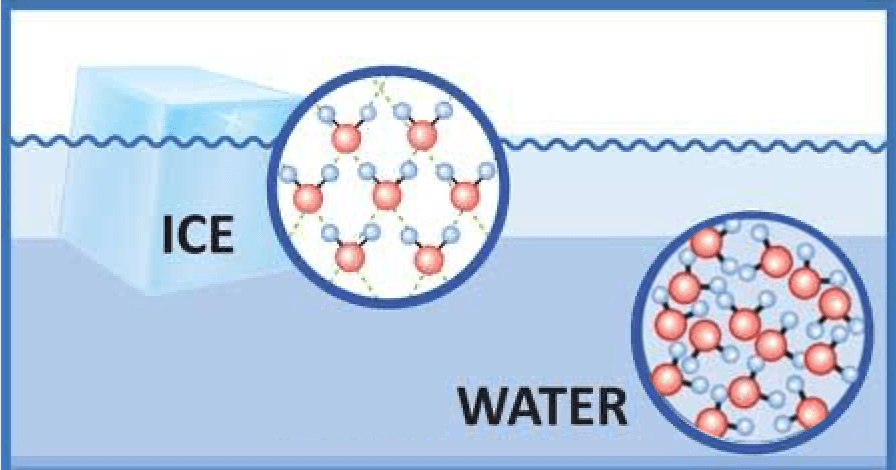
We call H2O molecules that are in a solid state:
Ice
Water
Steam/Vapor
2.
Multiple Choice

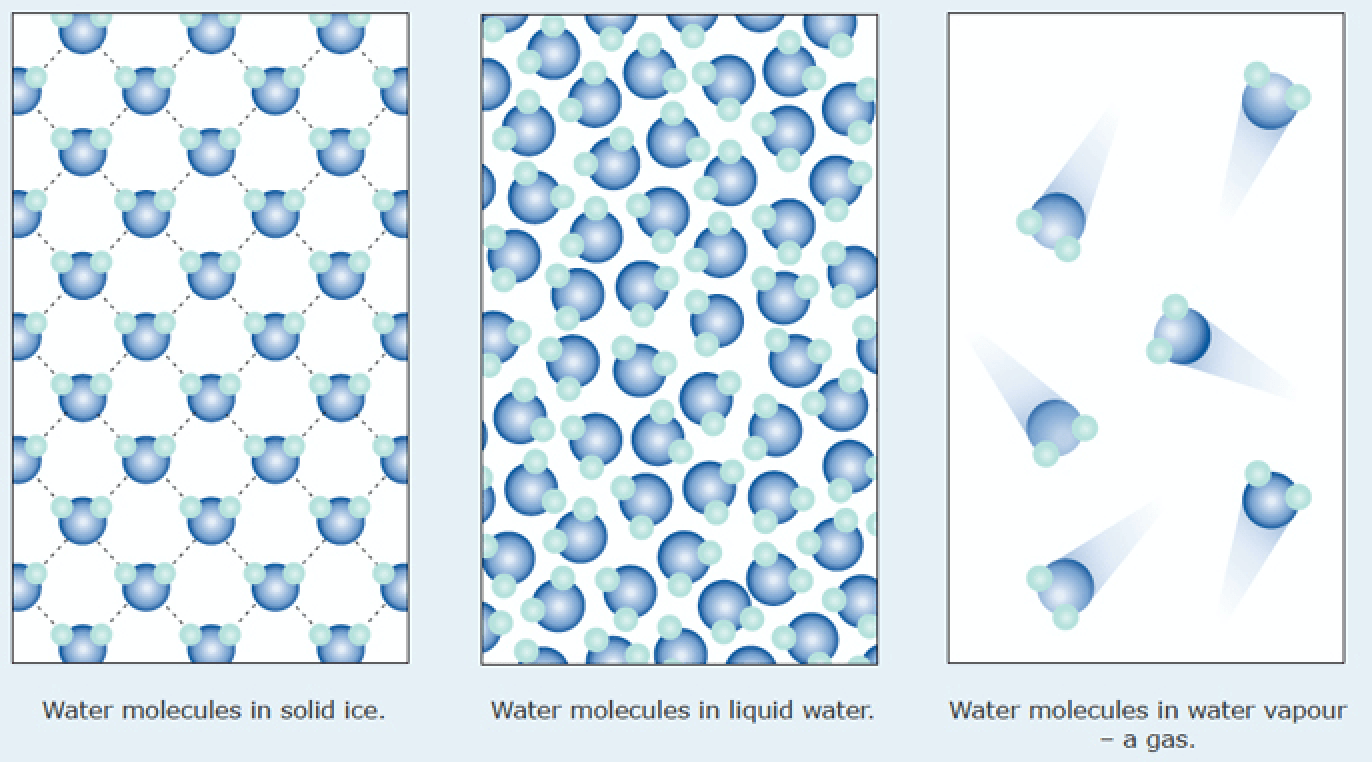
We call H2O molecules that are in a gaseous state:
Ice
Water
Steam or Vapor
3.
Multiple Choice
The molecules in a liquid move:
A lot and pretty fast.
A medium amount, they slide and glide.
Just a little.
Not at all.
4.
Multiple Choice

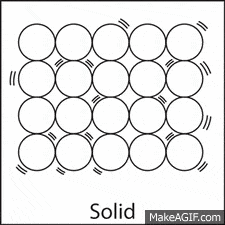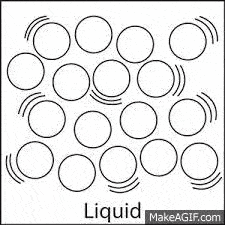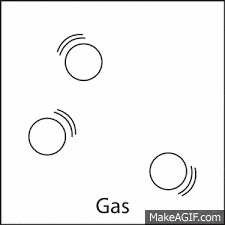
The molecules in a solid move:
A lot and pretty fast.
A medium amount, they slide and glide.
Just a little.
Not at all.
5.
Multiple Choice
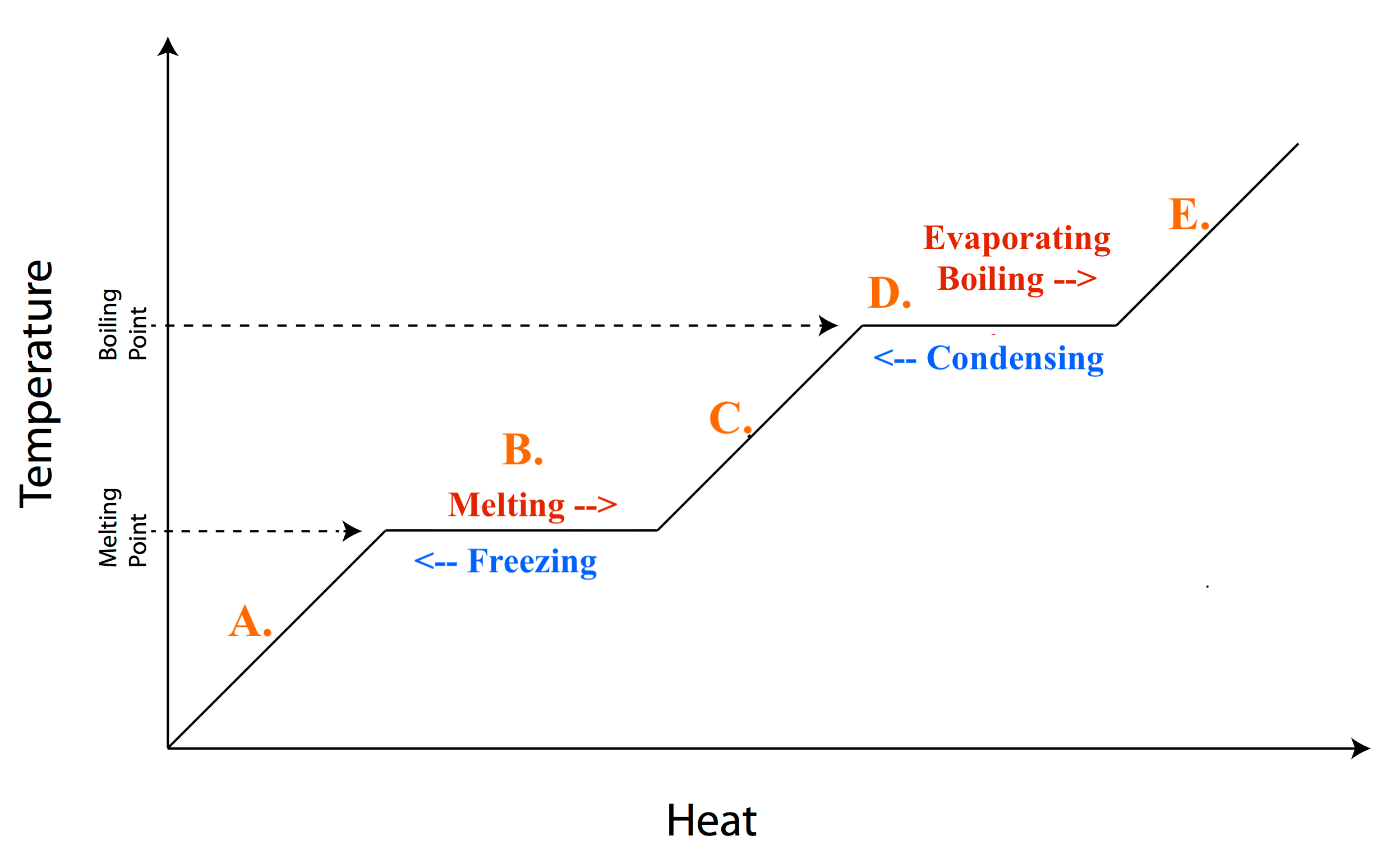
If the H2O molecules in ice are heated, they will start to:
Move faster.
Move slower.
6.
Multiple Select

If thermal energy increases:
(Check ALL that apply.)
The temperature gets hotter.
The temperature gets colder.
The molecules move slower.
The molecules move faster.
There is MORE energy overall.

Explore this activity with a free account
Find a similar activity
Create activity tailored to your needs using
.svg)

Properties of Matter
•
5th Grade

States of Matter
•
8th Grade

Physical Properties of Matter
•
5th Grade

Conservation of Matter
•
3rd - 5th Grade

States of Matter
•
3rd - 4th Grade

States of Matter
•
6th Grade

States of Matter
•
6th - 9th Grade