Fractional Distillation & Cracking Process
Assessment
•
Elanie Junita
•
Chemistry
•
10th - 11th Grade
•
159 plays
•
Hard
Improve your activity
Higher order questions
Match
•
Reorder
•
Categorization
.svg)
actions
Add similar questions
Add answer explanations
Translate quiz
Tag questions with standards
More options
25 questions
Show answers
1.
Multiple Choice
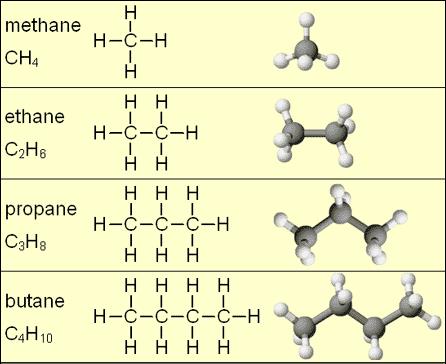
Fractional distillation separates crude oil based on the different __________ of the molecules in the mixture
melting point
boiling point
freezing point
chemical reactivity
2.
Multiple Choice
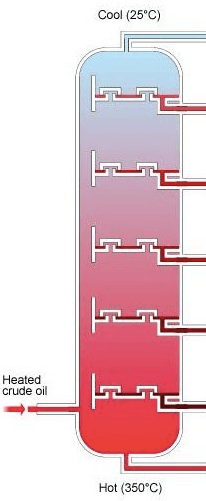
Molecules collected at the bottom of the fractionating column tend to be...
more viscous, less flammable and less volatile
less viscous, more flammable and more volatile
more viscous, more flammable and less volatile
less viscous, less flammable and more volatile
3.
Multiple Choice
Molecules collected at the top of the fractionating column tend to be...
more viscous, less flammable and less volatile
less viscous, more flammable and more volatile
more viscous, more flammable and less volatile
less viscous, less flammable and more volatile
4.
Multiple Choice
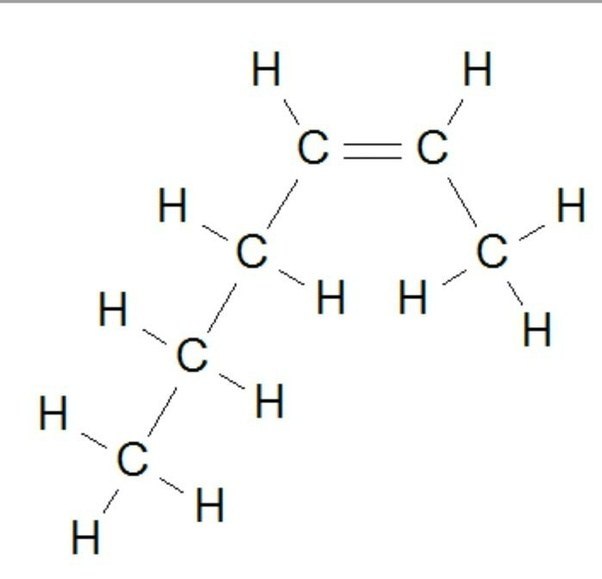
Name the following hydrocarbon
butane
hexane
heptane
hexene
5.
Multiple Choice
When testing for alkenes (unsaturation) bromine water turns from...
orange --> blue/black
colourless --> pink
orange --> colourless
orange --> clear
6.
Multiple Choice
a) Inner molecular forces
b) Outer molecular forces
c) Intermolecular forces
d) Intramolecular forces

Explore this activity with a free account
Find a similar activity
Create activity tailored to your needs using
.svg)

Hydrocarbons
•
11th - 12th Grade

Alkanes
•
12th Grade

Carbon Compound
•
8th Grade

Fractional Distillation & Cracking Process
•
10th - 11th Grade

Alkane And Alkene
•
7th - 11th Grade

Alcohols
•
1st Grade

Calorimetry
•
12th Grade

Isomers
•
12th Grade