
Identifying Redox Reactions
Assessment
•
HepChem HepChem
•
Chemistry
•
9th - 12th Grade
•
1K plays
•
Medium
Improve your activity
Higher order questions
Match
•
Reorder
•
Categorization
.svg)
actions
Add similar questions
Add answer explanations
Translate quiz
Tag questions with standards
More options
15 questions
Show answers
1.
Multiple Choice
Increased
Decreased
Remained Constant
Varies Randomly
2.
Multiple Choice
2CaO--> 2Ca + O2
Oxidized
Reduced
3.
Multiple Choice
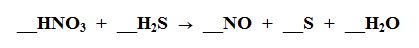
What element is being Oxidized?
N+5
H+1
O-2
S-2
4.
Multiple Choice
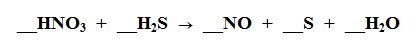
What element is being Reduced?
N+5
H+1
O-2
S-2
5.
Multiple Choice
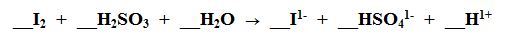
What element is being Oxidized?
I0
H+1
S+4
O-2
6.
Multiple Choice
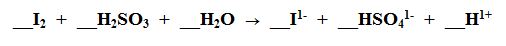
What element is being reduced?
I0
H+1
S+4
O-2

Explore this activity with a free account
Find a similar activity
Create activity tailored to your needs using
.svg)

Molar Mass
•
10th Grade - University

Redox Reactions
•
9th - 10th Grade

Naming Ionic Compounds
•
10th - 12th Grade

Naming Compounds
•
10th - 11th Grade

Element Symbols and Names
•
8th - 12th Grade

Moles and Mole Conversions
•
9th - 12th Grade

Ionic Bonding
•
8th - 12th Grade

Molarity
•
10th - 12th Grade