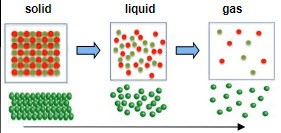
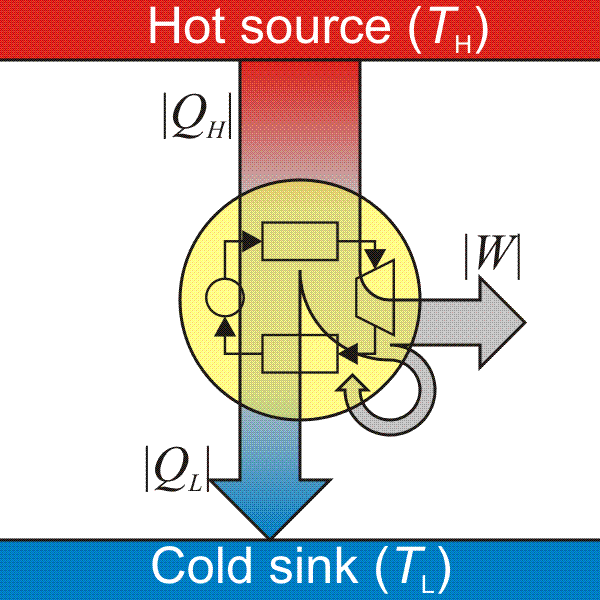
Laws of Thermodynamics
Assessment
•

Macky Mac
•
Physics
•
10th - 12th Grade
•
131 plays
•
Medium
Improve your activity
Higher order questions
Match
•
Reorder
•
Categorization
.svg)
actions
Add similar questions
Add answer explanations
Translate quiz
Tag questions with standards
More options
30 questions
Show answers
1.
Multiple Choice
What is thermodynamics?
study of heat and work
study of momentum
study of atoms
study of light
2.
Multiple Choice
true
false
3.
Multiple Choice
increase
decrease
stay constant
fluctuate wildly
4.
Multiple Choice

Match the temperature of the surrounding environment.
Always be colder than the surrounding environment.
Become warmer than the surrounding environment.
Never change temperature.
5.
Multiple Choice
created
destroyed
conserved
created and destroyed
6.
Multiple Choice
How many laws of thermodynamics are there?
5
4
3
2
1

Explore this activity with a free account
Find a similar activity
Create activity tailored to your needs using
.svg)

Heat Transfer
•
6th - 10th Grade

Heat and Temperature
•
5th - 8th Grade

Specific Heat
•
8th - 10th Grade

Thermal Energy
•
9th - 12th Grade

Thermodynamics
•
12th Grade

Convection, Conduction, Radiation
•
6th - 8th Grade

Heat and Temperature
•
8th - 9th Grade

Acceleration
•
10th Grade