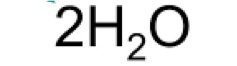Writing Chemical Equations Practice
Assessment
•
AMANDA M BRAMLETT
•
Chemistry
•
10th Grade
•
577 plays
•
Medium
Improve your activity
Higher order questions
Match
•
Reorder
•
Categorization
.svg)
actions
Add similar questions
Add answer explanations
Translate quiz
Tag questions with standards
More options
30 questions
Show answers
1.
Multiple Choice
Which is the correct chemical equation for the following: Solid Zn reacts with silver (I) chloride to produce Zinc (II) chloride and silver metal
Zn + AgCl → ZnCl2 + Ag
ZnCl2 + Ag → Zn + AgCl
ZnCl2 + Ag2 → Zn + AgCl
Zn2Cl + Ag → Zn + AgCl
2.
Multiple Choice
Which is the correct chemical equation for the following: Barium Chloride combined with sodium sulfide yields barium sulfide and sodium chloride
BaCl + NaS → BaS+ NaCl
BaCl2 + Na2S → BaS+ NaCl
BaS+ NaCl → BaCl + NaS
BaCl2 + Na2S → BaS+ NaCl2
3.
Multiple Choice
NaO
NaO2
Na2O
Na2O2
4.
Multiple Choice
beryllium chlorine
beryllium II chloride
beryllium chloride
beryllium dichloride
5.
Multiple Choice
Fluorine
Nitrogen
Boron
Bromine
6.
Multiple Choice
___________________ are the elements or compounds that are formed in a chemical reaction.
Polymers
Bonds
Reactants
Products

Explore this activity with a free account
Find a similar activity
Create activity tailored to your needs using
.svg)

Molar Mass
•
10th Grade - University

Redox Reactions
•
9th - 10th Grade

Naming Ionic Compounds
•
10th - 12th Grade

Naming Compounds
•
10th - 11th Grade

Element Symbols and Names
•
8th - 12th Grade

Moles and Mole Conversions
•
9th - 12th Grade

Ionic Bonding
•
8th - 12th Grade

Molarity
•
10th - 12th Grade